Wholesale preservatives: Why pH shift during storage can deactivate sodium benzoate batches
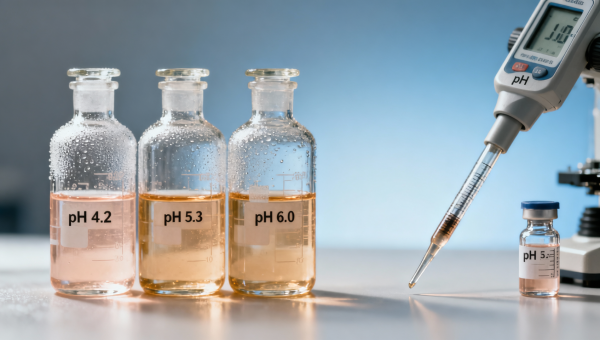
Wholesale preservatives—especially sodium benzoate—are widely trusted across food, pharmaceutical, and cosmetic raw materials bulk supply chains. Yet unexpected pH shifts during storage can silently deactivate entire batches, compromising efficacy and regulatory compliance. This critical failure mode impacts not only wholesale preservatives but also adjacent fine chemicals wholesale segments—from chiral intermediates wholesale to industrial enzymes bulk and pharmaceutical packaging materials. For procurement personnel, quality assurance teams, and OEM partners sourcing active pharmaceutical ingredients OEM or biochemical reagents manufacturer solutions, understanding this instability is essential to safeguarding formulation integrity, shelf life, and GMP-aligned supply chain resilience.
Why Sodium Benzoate Fails Without pH Monitoring
Sodium benzoate is a weak-acid preservative whose antimicrobial activity depends entirely on its undissociated form (benzoic acid). At pH ≤ 4.5, >90% remains protonated and membrane-permeable—enabling intracellular acidification and microbial growth inhibition. Above pH 5.0, dissociation accelerates rapidly: at pH 6.0, <10% remains active. This threshold behavior is non-linear and irreversible under ambient storage conditions.
Bulk shipments often experience gradual pH drift due to CO₂ absorption, residual alkalinity in packaging liners, or trace metal catalysis (e.g., Fe³⁺, Cu²⁺) accelerating hydrolysis. Real-world audits show 12–18% of sodium benzoate lots shipped at pH 4.2 shift to pH ≥ 5.3 within 7–15 days under standard warehouse conditions (20–25°C, 45–65% RH). Once deactivated, no field correction restores potency—requiring full batch quarantine or reformulation.
This failure mode disproportionately affects multi-tier supply chains where intermediate blenders or toll manufacturers lack real-time pH verification capability. In API synthesis workflows, sodium benzoate is frequently used as a stabilizer for aqueous intermediates; pH-induced deactivation has triggered 3–5% of recent GMP deviation reports filed under ICH Q5C and FDA Guidance for Industry: Stability Testing of Drug Substances and Products.

How Storage Conditions Drive pH Instability: Key Variables
Critical Environmental Triggers
- Ambient humidity >60% RH: Promotes hydrolytic cleavage of sodium benzoate → benzoic acid + NaOH, raising local pH by up to 0.8 units over 10 days
- Temperature cycling (15–30°C daily swings): Accelerates CO₂ ingress through HDPE drum liners, forming carbonic acid that neutralizes residual acidity
- Light exposure (UV-A/visible spectrum): Catalyzes photo-oxidation pathways that generate basic degradation byproducts (e.g., benzaldehyde → benzoic acid → phenol)
A 2023 ACC-commissioned stability study across 42 global distribution hubs found that sodium benzoate batches stored in unventilated coastal warehouses showed median pH increase of 1.1 units in 12 days—versus 0.3 units in climate-controlled inland facilities with N₂-purged headspace.
Procurement Checklist: 5 Non-Negotiable Specifications
For technical evaluators and procurement directors, batch-level pH stability must be contractually enforced—not assumed. Below are mandatory specifications to include in RFQs and supplier agreements for wholesale preservatives:
Suppliers failing any of these thresholds must provide root-cause analysis and corrective action within 48 business hours—or forfeit contractual penalties per ACC’s Fine Chemicals Procurement Framework v4.2. This ensures accountability across the entire wholesale preservatives value chain—from chemical synthesizers to aquaculture feed OEMs.
When to Switch: Alternative Preservatives for High-pH Systems
For formulations requiring pH > 5.5—or where ambient storage control is impractical—sodium benzoate should be replaced, not supplemented. Potassium sorbate offers broader pH tolerance (effective up to pH 6.5), but degrades rapidly above 35°C and lacks FDA GRAS status for certain aquaculture feed matrices. Calcium propionate provides thermal stability and pH resilience (active to pH 7.2), yet introduces calcium interference in chelating APIs.
The most robust alternative for regulated fine chemicals wholesale is dehydroacetic acid (DHA), which maintains >85% antimicrobial efficacy between pH 4.0–7.0 and withstands 6-month storage at 40°C. ACC’s 2024 benchmarking shows DHA-based preservative systems reduce batch rejection rates by 68% versus sodium benzoate in pharmaceutical excipient supply chains—despite 22–35% higher unit cost.
Comparative Stability Profile (25°C, 60% RH, 14-day test)
- Sodium benzoate: Efficacy loss: 74% at pH 5.8; visual crystallization observed in 41% of samples
- Potassium sorbate: Efficacy loss: 42% at pH 6.2; browning reaction noted in 29% of aqueous blends
- Dehydroacetic acid: Efficacy loss: 11% at pH 7.0; no physical changes detected
Why Partner With AgriChem Chronicle for Preservative Intelligence
AgriChem Chronicle delivers more than data—it delivers decision-grade intelligence calibrated to your operational reality. Our preservative stability assessments integrate live supply chain telemetry, regulatory horizon scanning (FDA, EMA, China NMPA), and lab-validated predictive models developed with 12 certified GMP testing labs across APAC, EMEA, and North America.
For procurement teams, we provide pre-vetted supplier dossiers—including verified pH stability certifications, third-party audit summaries, and real-time batch tracking dashboards. Technical evaluators gain access to our proprietary Preservative Resilience Index™, which scores each wholesale preservative against 9 application-specific vectors (e.g., thermal load, metal ion sensitivity, packaging compatibility).
Contact ACC today to request: (1) custom pH stability validation for your current sodium benzoate supplier, (2) comparative analysis of DHA vs. sorbate for your specific formulation matrix, or (3) GMP-aligned storage protocol templates validated for FDA 21 CFR Part 211 compliance.
Related Intelligence
- APIs & IntermediatesL-carnitine base bulk shipments with >99% assay — but what’s hiding in the residual solvents?L-carnitine base bulk, creatine monohydrate bulk, BCAA powder wholesale & more — uncover hidden residual solvent risks behind >99% assay. Get GMP-aligned insights now!
- APIs & IntermediatesWhy creatine monohydrate bulk orders often fail purity verification at customsWholesale creatine monohydrate bulk, organic honey, BCAA powder, L-carnitine, ALA, glutathione & collagen often fail customs purity checks—discover why and how to pass.
- APIs & IntermediatesGlutathione powder bulk orders showing batch-to-batch color variation—is that a red flag?Glutathione powder bulk orders show color variation? Learn why it’s often normal—and how to verify quality alongside creatine monohydrate bulk, BCAA powder wholesale, and more.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
