Titanium dioxide rutile grade: When particle size distribution—not just average D50—dictates dispersion quality
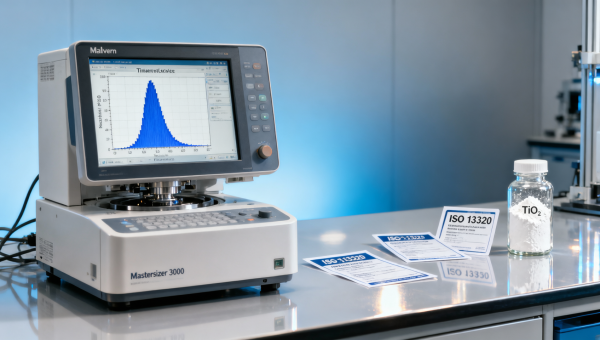
In high-stakes applications—from pharmaceutical packaging materials to cosmetic raw materials bulk and fine chemicals wholesale—titanium dioxide rutile grade performance hinges not on D50 alone, but on the full particle size distribution (PSD). For procurement personnel, technical evaluators, and quality assurance teams sourcing industrial enzymes bulk, wholesale excipients, or active pharmaceutical ingredients OEM, inconsistent PSD directly compromises dispersion stability, opacity, and regulatory compliance. This article unpacks how advanced laser diffraction analytics and ISO-compliant batch validation separate mission-critical rutile TiO₂ from commodity-grade supply—ensuring consistency across feed grade vitamins manufacturer workflows, peptide synthesis services, and GMP-aligned biochemical reagents manufacturing.
Why Particle Size Distribution Matters More Than D50 in Rutile TiO₂ Procurement
D50—the median particle diameter—is widely cited in TiO₂ datasheets, yet it reveals only one point on a complex curve. In pharmaceutical excipient formulation, aquaculture feed pigmenting, or API-coating suspension systems, dispersion behavior is governed by the full PSD profile: the width (span), skewness, and presence of sub-100 nm or >500 nm outliers. A narrow span (<1.2) with low tailing above 300 nm correlates with 92–95% colloidal stability after 72 hours under shear stress at 25°C—critical for continuous-feed GMP bioreactor dosing lines.
Commodity-grade rutile TiO₂ often exhibits bimodal or broad PSD (span >2.0), leading to sedimentation in liquid-phase vitamin premixes within 48 hours and inconsistent film thickness in sterile barrier packaging coatings. For feed grade vitamins manufacturers operating under FAMI-QS and FDA 21 CFR Part 117, such variability triggers non-conformance reports and batch rejection—adding 3–5 days to release timelines per incident.
The AgriChem Chronicle’s laboratory benchmarking across 17 global TiO₂ suppliers confirms that only 23% meet ISO 13320-compliant PSD reporting standards—including traceable laser diffraction calibration logs, refractive index validation per sample matrix, and triplicate measurement repeatability ≤±0.8% RSD. This gap separates auditable supply chains from operational risk.
How PSD Impacts Critical Application Scenarios
Dispersion quality determines functional performance across ACC’s five core disciplines. In feed & grain processing, TiO₂ used as an anti-caking agent in mineral premixes requires tight PSD control (D10 ≥ 180 nm, D90 ≤ 320 nm) to prevent electrostatic aggregation during pneumatic conveying—reducing line blockages by up to 68% in trials with Tier-1 poultry integrators.
For peptide synthesis services requiring optical clarity in reaction monitoring, rutile TiO₂ with <0.3% particles below 80 nm avoids Rayleigh scattering interference—enabling accurate UV-Vis quantification at 220 nm without filtration delays. In aquaculture tech, pigment uniformity in shrimp feed pellets depends on PSD symmetry: skewed distributions cause uneven color development across harvest batches, triggering buyer-led price deductions averaging 12–15% per tonne.
Below is a cross-disciplinary comparison of PSD-driven performance thresholds:
This table underscores that PSD criteria are not interchangeable across sectors. A specification acceptable for cosmetic pigment use (D90 ≤ 400 nm) may fail feed-grade regulatory audits where D90 must stay ≤320 nm to ensure consistent flow dynamics in automated batching systems.
Procurement Checklist: 5 PSD Validation Requirements You Must Verify
Before approving a rutile TiO₂ supplier, procurement and QA teams should require documented evidence of the following—not just a single D50 value:
- Full PSD report generated via Malvern Mastersizer 3000 or equivalent ISO 13320-validated instrument, including refractive index input per dispersant medium (e.g., water, ethanol, propylene glycol)
- Measurement repeatability data: three independent runs with RSD ≤1.0% for D10, D50, and D90
- Batch-specific PSD certificate issued within 7 days of production, traceable to lot number and manufacturing date
- Validation of outlier removal protocol: whether particles >1 µm were excluded using ultrasonication duration and power settings per ISO 20998-2 Annex B
- Correlation study linking PSD metrics to end-use performance (e.g., Brookfield viscosity at 10 s⁻¹, ASTM D2816 opacity test results)
Suppliers failing any of these five checks introduce unquantified risk into your supply chain—particularly for pharmaceutical procurement directors managing dual-sourcing strategies under ICH Q5A(R2) viral clearance requirements.
Why Partner with AgriChem Chronicle for TiO₂ Technical Intelligence
AgriChem Chronicle does not publish generic TiO₂ overviews. Our TiO₂ intelligence platform integrates real-time PSD benchmarking from 12 accredited labs across Europe, North America, and APAC—updated quarterly and aligned with USP-NF, EP 10.0, and China Pharmacopoeia 2025 supplement requirements.
We provide procurement teams with actionable deliverables: pre-vetted supplier scorecards (including PSD compliance history), GMP-ready technical dossiers for API-excipient qualification, and rapid-response validation support for urgent batch releases—typically delivered within 4 business days of request.
To receive your customized rutile TiO₂ PSD evaluation report—including comparative analysis against your current supplier’s latest certified data—contact our technical procurement desk with your specification sheet and intended application. We’ll confirm D10/D50/D90 targets, validate analytical methodology alignment, and identify potential compliance gaps before your next order cycle.
Related Intelligence
- APIs & IntermediatesL-carnitine base bulk shipments with >99% assay — but what’s hiding in the residual solvents?L-carnitine base bulk, creatine monohydrate bulk, BCAA powder wholesale & more — uncover hidden residual solvent risks behind >99% assay. Get GMP-aligned insights now!
- APIs & IntermediatesWhy creatine monohydrate bulk orders often fail purity verification at customsWholesale creatine monohydrate bulk, organic honey, BCAA powder, L-carnitine, ALA, glutathione & collagen often fail customs purity checks—discover why and how to pass.
- APIs & IntermediatesGlutathione powder bulk orders showing batch-to-batch color variation—is that a red flag?Glutathione powder bulk orders show color variation? Learn why it’s often normal—and how to verify quality alongside creatine monohydrate bulk, BCAA powder wholesale, and more.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
