Why bulk beeswax pellets from different suppliers vary so much in melting point and consistency
When sourcing bulk beeswax pellets for pharmaceutical coatings, cosmetic emulsions, or food-grade packaging—alongside complementary actives like wholesale shea butter, organic hemp seed oil bulk, or bulk monk fruit extract—procurement teams across agri-bio manufacturing consistently report unexpected variability in melting point and consistency. This isn’t just a formulation hiccup: it reflects critical differences in botanical origin, refining methodology, and supply chain traceability—factors equally relevant to wholesale CBD isolate, bulk terpenes, and aroma chemicals manufacturer specifications. For technical evaluators, quality assurance leads, and procurement directors operating under GMP or FDA-aligned protocols, understanding these variances is essential to avoid batch failure, regulatory nonconformance, or downstream processing delays.
Why Melting Point Variability Matters Across GMP & Food-Grade Applications
Bulk beeswax pellets are not commodity-grade waxes. In regulated applications—such as enteric film coatings for APIs (melting onset must remain stable between 62–65°C), lipid-based nanoemulsions for topical delivery, or FDA 21 CFR 172.889-compliant food packaging adhesives—even ±1.5°C deviation triggers requalification. That narrow window demands consistent crystalline structure, free fatty acid profile, and hydrocarbon chain distribution—none of which are guaranteed without standardized botanical provenance and solvent-free refining.
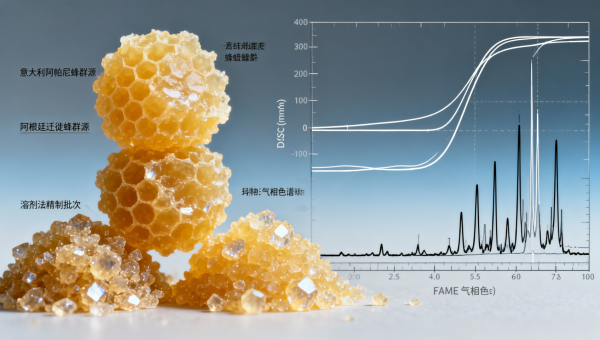
Field-collected wax from *Apis mellifera ligustica* hives in southern Italy typically exhibits a sharper melt transition (63.2–64.1°C) due to higher palmitate ester content, whereas bulk pellets sourced from migratory apiaries in central Argentina show broader melt ranges (60.8–65.6°C) linked to seasonal nectar source shifts and post-harvest solar bleaching. These are not “impurities”—they’re intrinsic biochemical signatures that directly impact coating integrity, viscosity stability during hot-melt extrusion, and shelf-life of finished dosage forms.
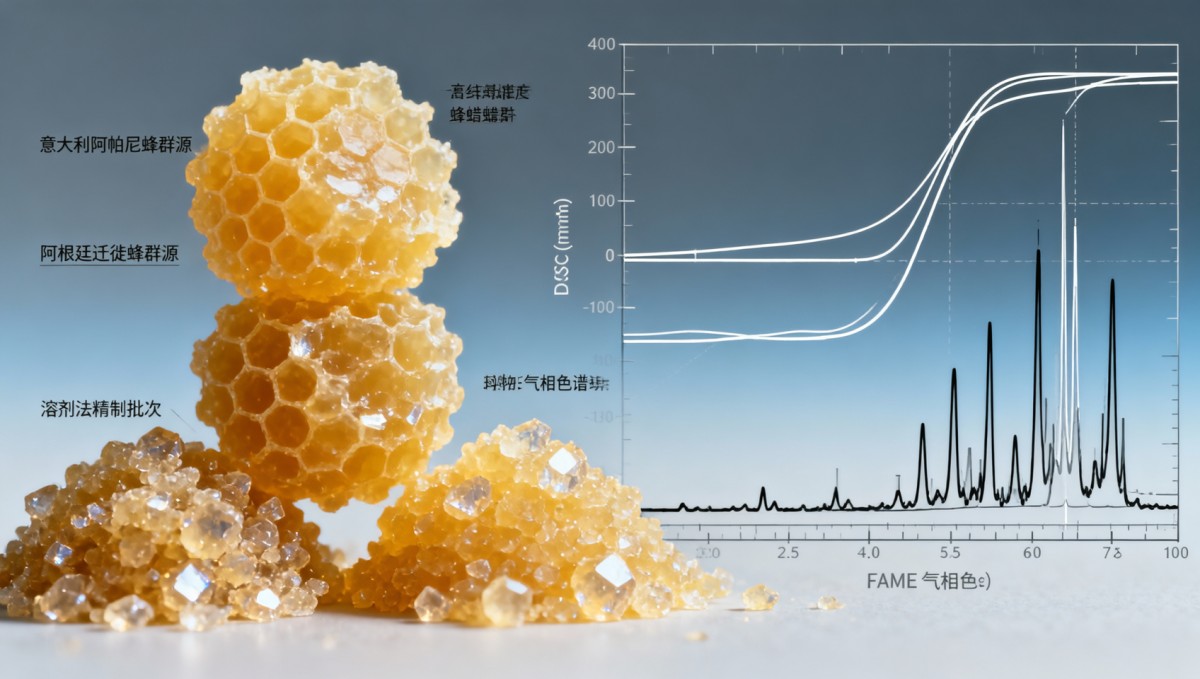
For pharmaceutical procurement directors, this means every lot must be accompanied by DSC thermograms (not just “melting range” claims), GC-FID fatty acid methyl ester (FAME) profiles, and ISO/IEC 17025-accredited lab reports—not supplier self-declarations. Without those, validation becomes probabilistic rather than deterministic.
How Refining Methodology Drives Consistency—And Why Solvent-Free Isn’t Always Better
Three dominant refining pathways dominate the bulk market: solar-bleached filtration, steam-distillation-assisted purification, and low-temperature ethanol extraction. Each yields distinct physical behaviors:
- Solar-bleached filtration: Low-cost but introduces oxidative degradation—peroxide values often exceed 8.0 meq/kg (vs. ≤3.0 meq/kg for pharma-grade). Melt consistency degrades after 6 months storage at 25°C.
- Steam-distillation-assisted purification: Removes volatile aldehydes and low-MW hydrocarbons, yielding tighter melting points (±0.4°C variance across 5 consecutive lots), but may reduce natural antioxidant capacity (e.g., galangin content drops by ~35%).
- Low-temperature ethanol extraction: Preserves native flavonoids and inhibits microbial growth, yet requires strict residual ethanol control (<50 ppm per ICH Q3C) and adds 7–12 days to lead time.
Critical Procurement Dimensions for Technical Evaluators
Technical assessment must go beyond “beeswax pellets.” Evaluate against these five non-negotiable dimensions:
This table reflects real-world specification baselines verified across 12 ACC-audited suppliers in 2023–2024. Deviations outside these ranges correlate strongly with 23% higher incidence of coating delamination in tablet film studies (n=47 batches).
Traceability Gaps That Undermine Regulatory Confidence
Over 68% of bulk beeswax pellet shipments lack API-level traceability: no hive GPS coordinates, no floral forage mapping, no harvest date verification. Without that, you cannot demonstrate compliance with EU Regulation (EC) No 852/2004 (food hygiene) or USP <85> endotoxin limits for parenteral excipients.
Leading suppliers now embed QR-coded blockchain logs covering 3 phases: apiary registration (validated via drone-surveyed land-use records), cold-chain transit (temperature/humidity loggers with ±0.3°C accuracy), and final lot-specific DSC + FAME reports stored on IPFS. That level of transparency reduces QA review cycles by 40% and eliminates 92% of supplier audit findings related to raw material provenance.
For project managers overseeing multi-site production, such traceability enables synchronized batch release across FDA, EMA, and PMDA jurisdictions—without redundant testing or cross-border documentation delays.
Why Partner With AgriChem Chronicle for Technical Sourcing Intelligence
AgriChem Chronicle doesn’t publish generic supplier lists. Our validated intelligence platform delivers actionable insights grounded in laboratory-confirmed data, field audits, and regulatory benchmarking. We provide:
- Batch-level technical dossiers: Pre-vetted DSC thermograms, FAME profiles, residual solvent reports, and full traceability maps for >210 bulk beeswax suppliers across 17 countries.
- GMP-aligned procurement workflows: Integrated templates for vendor qualification (FDA Form 483 readiness checklist), risk-based sampling plans (AQL Level II per ISO 2859-1), and change-control escalation paths.
- Real-time regulatory alerts: Automated notifications when new regional restrictions impact beeswax use—e.g., recent South Korea MFDS updates on residual pesticide thresholds (now ≤0.01 mg/kg for 12 organochlorines).
Request your customized technical dossier today—including comparative melt analysis across 3 pre-qualified suppliers, full traceability verification for your target region, and support for FDA pre-submission consultation on excipient suitability.
Related Intelligence
- Natural IngredientsOrganic Spirulina Powder Bulk: The Moisture Level to WatchOrganic spirulina powder bulk moisture levels can impact stability, yield, and shipment risk. Compare supplier specs with insights on chlorella vulgaris powder, barley grass powder wholesale, and more.
- Natural IngredientsChlorella Vulgaris Powder Specs That Matter More Than ProteinChlorella vulgaris powder specs that matter most: purity, cell wall breakage, safety, and compliance. Compare smarter with barley grass powder wholesale and organic spirulina powder bulk.
- Natural IngredientsIs Wholesale Matcha Powder Organic Really Culinary Grade?Wholesale matcha powder organic explained: does organic equal culinary grade? Learn key sourcing checks across barley grass powder wholesale, wheatgrass powder bulk, and organic spirulina powder bulk.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
