ANVISA Extends Custom Synthesis Traceability to 3rd-Tier Suppliers
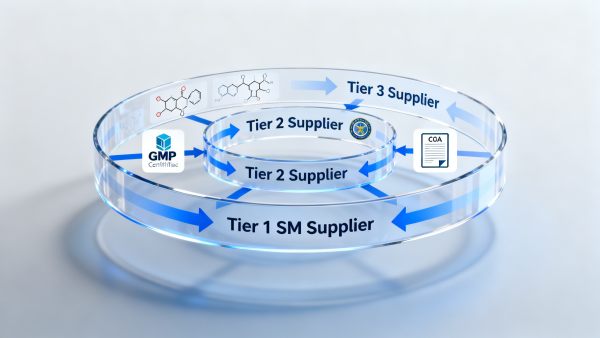
On April 12, 2026, Brazil’s National Health Surveillance Agency (ANVISA) updated its Guidelines for Importation of Active Pharmaceutical Ingredients, mandating full traceability of starting materials (SM) for custom-synthesized APIs back to the third-tier upstream supplier. This development directly affects API importers, contract manufacturers, and global suppliers serving the Brazilian pharmaceutical market—and signals tightening regulatory expectations for supply chain transparency in Latin America.
Event Overview
On April 12, 2026, ANVISA published an update to its Guidelines for Importation of Active Pharmaceutical Ingredients. The revision requires importers of custom-synthesized active pharmaceutical ingredients (APIs) to submit complete溯源 documentation for starting materials—including certificates of analysis (COA), process flow diagrams, and GMP compliance statements—covering all suppliers up to the third tier upstream from the immediate SM supplier. The requirement becomes mandatory on October 1, 2026. During the transition period (April 12–September 30, 2026), importers may submit a ‘Traceability Commitment Letter’, provided it is accompanied by a verifiable third-party audit plan.
Which Subsectors Are Affected
Direct Trade Enterprises (API Importers into Brazil)
These entities are the primary obligated parties under the new rule. They must now collect, validate, and retain documentation not only from their direct SM suppliers but also from two additional tiers upstream—potentially including raw chemical producers or specialty intermediate manufacturers. Impact manifests in increased due diligence burden, longer import clearance lead times, and higher risk of customs hold or rejection if documentation is incomplete or inconsistent.
Raw Material Procurement Entities (Global SM Sourcing Teams)
Pharmaceutical companies with centralized procurement functions—especially those sourcing SMs from Asia or Europe for use in custom synthesis—will face new coordination requirements. Their internal systems must now map and verify multi-tier supplier relationships, assess audit readiness across tiers, and align documentation formats (e.g., COA templates, GMP declarations) with ANVISA’s expectations. Failure to standardize upstream data collection may delay API registration or batch release in Brazil.
Contract Development and Manufacturing Organizations (CDMOs)
CDMOs engaged in custom API synthesis for Brazilian clients must proactively disclose and document their SM supply chains beyond Tier 1. Since many CDMOs rely on subcontracted intermediates or toll-manufactured precursors, this rule compels them to formalize agreements with Tier 2 and Tier 3 partners—including audit rights and data-sharing clauses. Absent such contractual clarity, CDMOs may be unable to fulfill client commitments or maintain ANVISA-compliant dossiers.
Supply Chain Verification & Compliance Service Providers
Third-party auditors, regulatory consultants, and supply chain mapping platforms will see increased demand for tiered supplier verification services—particularly those capable of validating GMP alignment and technical consistency across multiple jurisdictions. However, the requirement for a ‘verifiable third-party audit plan’ during the transition phase implies ANVISA expects specificity: scope, timeline, methodology, and auditor credentials—not just generic intent.
What Relevant Companies or Practitioners Should Focus On Now
Monitor Official Implementation Guidance and Clarifications
ANVISA has not yet published detailed definitions of ‘Tier 1/2/3 supplier’, nor clarified whether ‘upstream’ refers to manufacturing steps or contractual relationships. Stakeholders should track upcoming Q&A documents or webinars from ANVISA’s Medicines Directorate—and avoid assuming interpretations until formally confirmed.
Map Current SM Supply Chains Against the Three-Tier Requirement
Importers and CDMOs should conduct an internal gap assessment: identify which current SMs originate from multi-tier structures, determine where documentation is missing (e.g., COAs from Tier 2), and prioritize remediation for high-volume or high-risk APIs. Mapping should distinguish between owned facilities, contracted sites, and toll manufacturers—each posing distinct verification challenges.
Prepare Transition-Period Documentation Strategically
The ‘Traceability Commitment Letter’ is permissible only if paired with a concrete third-party audit plan. That plan must specify which tiers will be audited, the audit standards applied (e.g., WHO-GMP, PIC/S), and expected completion dates. Vague or delayed timelines may invalidate the commitment—and expose submissions to rejection.
Engage Upstream Suppliers Early on Documentation Standards
Many Tier 2 and Tier 3 suppliers—especially small- to mid-sized chemical producers—may lack standardized COAs or formal GMP declarations. Rather than waiting for ANVISA queries, importers should initiate supplier outreach now to co-develop acceptable documentation templates and clarify expectations around process flow diagram detail (e.g., inclusion of solvents, reagents, critical quality attributes).
Editorial Perspective / Industry Observation
From an industry perspective, this update is less a sudden enforcement shift and more a formalization of an emerging regional expectation: that API supply chain integrity extends beyond the immediate vendor. Analysis来看, ANVISA’s move aligns with broader trends seen at EDQM and Health Canada—where increasing emphasis is placed on understanding material genealogy, not just final product testing. It is currently more accurate to interpret this as a strong regulatory signal than an immediate operational outcome: the six-month transition window, coupled with the allowance of commitment-based submissions, suggests ANVISA anticipates capacity-building needs across the ecosystem. Observation来看, the focus on ‘custom synthesis’—rather than all APIs—indicates targeted scrutiny of higher-risk, lower-volume, chemically complex substances where impurity profiles and route-dependent genotoxic risks are harder to control without deep supply chain insight.
Conclusion
This update does not represent a standalone policy change but rather a calibrated escalation in supply chain accountability for a specific, high-scrutiny segment of the API trade. Its significance lies not in novelty—multi-tier traceability has been discussed globally for years—but in its codification within a major emerging-market regulator’s binding guidance. For stakeholders, the most rational interpretation is that ANVISA is institutionalizing expectations already embedded in ICH Q5A(R2) and WHO TRS 1033, now adapted to local enforcement realities. Preparedness—not panic—is the appropriate response.
Information Sources
Main source: ANVISA Official Notice No. 47/2026, dated April 12, 2026, updating the Guidelines for Importation of Active Pharmaceutical Ingredients.
Points requiring ongoing observation: ANVISA’s forthcoming definitions of supplier tiers, acceptance criteria for third-party audit plans, and potential issuance of illustrative case studies or FAQs.
Related Intelligence
- Custom SynthesisANVISA Extends Custom Synthesis Traceability to 3rd-Tier SuppliersANVISA Extends Custom Synthesis Traceability to 3rd-Tier Suppliers—key update for API importers, CDMOs & global suppliers entering Brazil. Act now.
- Custom SynthesisANVISA Extends Custom Synthesis API Traceability to 3-Tier Upstream SuppliersANVISA extends Custom Synthesis API traceability to 3-tier upstream suppliers—key for pharma importers, CMOs & supply chain teams. Act now to avoid suspensions.
- Custom SynthesisANVISA Expands Custom Synthesis Review to 3-Tier Starting Material TraceabilityANVISA's new 3-tier traceability rule for custom synthesis imports demands full upstream documentation—act now to avoid delays, rejections, and compliance risks in Brazil.
The Morning Broadsheet
Daily chemical briefings, market shifts, and peer-reviewed summaries delivered to your terminal.
